Medical Devices Regulation (EU) 2017/745 - MDR
As a trusted notified body, DNV can help you navigate the complexity of the EU’s Medical Device Regulation and get access to the EU market.
Medical Devices Regulation (EU) 2017/745 - MDR
On 5 May 2017, two new Regulations on medical devices were published, and they entered into force on 25 May 2017. The Medical Devices Regulation (MDR) date of application is 26 May 2021 meaning compliance is mandatory to be able to place Medical Devices on the European market from this date, unless the transition arrangements allows the continued placing of devices on the market. In vitro Diagnostic Medical Devices Regulation (IVDR) applies from 26 May 2022.
Products intended for the UK market must be compliant with the new product marking UKCA launched by the UK government on 1 January 2021. For medical devices, the UKCA transition period is currently set to 30 June 2023.
What is Medical Devices Regulation (EU) 2017/745 - MDR
Medical Devices Regulation (EU) 2017/745 has replaced the Medical Device Directive (MDD) and the Active Implantable Medical Device Directive (AIMD), whereas the IVDR will replace the In vitro Diagnostic Directive (IVDD). Both regulations bring a series of important improvements to conformity assessment for medical devices with the intention to:
- Improve the quality, safety and reliability of medical devices placed on the European market.
- Strengthen transparency of information related to medical devices for consumers and practitioners.
- Enhance vigilance and market surveillance of devices in use.
The extent to which these changes will affect your operations will depend on the type of device manufactured and the role you hold (manufacturer, importer, authorized representative). It is however anticipated that in most cases significant changes must be made to compliance process, quality management system, and technical documentation and successfully implemented, and certified as relevant to device classes and roles of your organization, before compliance with the regulations can be achieved.
See an overview of some of the key changes here.
Continued validity of MDD certificates
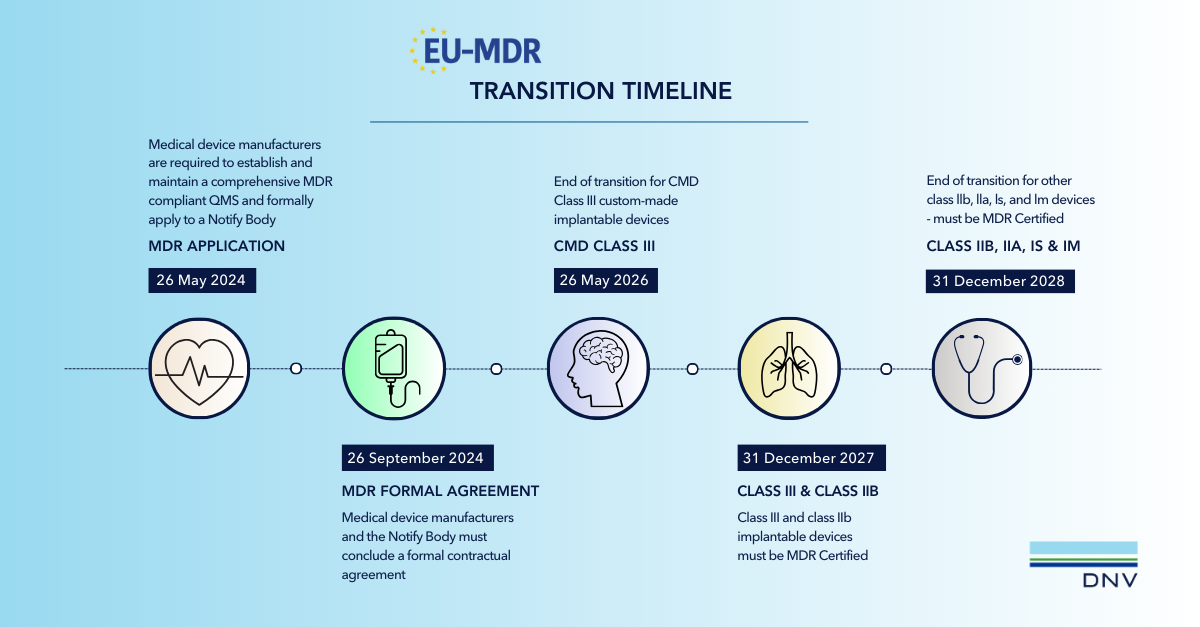
MDD certificates will be valid until their original expiry date or 26 May 2024, whichever is sooner. This is provided that compliance is maintained, certain elements of the MDR are fulfilled and the Notified body performs surveillance activities.
Thus manufacturers can continue to put MDD certified devices on the market up to and including 26 May 2024. It is important to note however that no significant changes may be or have been made to MDD certified devices after 26 May 2020. If a significant change is required, the device certificate must be migrated to MDR.
Migration of MDD certified devices to MDR
The changes to the MDR and IVDR are often seen as a revision of the requirements. In reality, it is a new regulation and should be treated as such. In general, the concepts and the requirements of the MDD and IVDD remain part of the new legislation but significant additional requirements have been added and changes made. There are a number of new requirements that manufacturers need to address before an application for certification to the MDR and IVDR can be made.
A new application for certification must be made by the legal manufacturer which will commence a new certification cycle, with validity up to 5 years. Manufacturers shall successfully complete a Notified Body review of technical files and an initial on-site audit activity to verify implementation before certification will be granted. The review of technical files is based on sampling, however this is not permitted for Class IIb implantable or Class III devices. Manufacturers shall ensure their technical files and design dossiers are MDR compliant prior to application.
MDR Certification Process
In order to support your migration to MDR, we have prepared the following visualization of the certification process and key information regarding the different types of activity. Note: this is intended as an illustration, not an exhaustive list of requirements.
Getting ready for certification
Whether migrating from MDD to MDR or just starting out, it is essential to first understand the regulation and requirements. As mentioned, there are substantial changes to compliant processes, the quality management system and technical documentation.
To help you understand the readiness of your MDR quality management system, take advantage of our free to use self-assessment tool. To register, please email us ready.support@dnv.com. The tool is accessible from our self-assessment micro-site: https://ready.dnv.com*.
Certification by a notified body is required to receive the CE marking on your device. Note that for products intended for the UK market, the UKCA will be required. The transition date is currently set to 30 June 2023.
DNV can support you throughout the journey, from training to getting ready for certification and the certification process itself to all market access standards and ISO 13485 (medical device quality management system).
DNV MDR designation
DNV is designated by the Norwegian Health Authorities and the European Commission as a Notified Body 2460 for Medical Devices Regulation (EU) 2017/745 (MDR). The designation is granted for all technology types applied for, including the highest risk Class III devices, ensuring continuity of European market access for our customers’ life-saving medical technology.
This consolidates the two previous MDD notifications for DNV GL Presafe and Presafe Denmark AS. The reason is to ensure we can provide a more effective service for our customers by combining our global technical resources (over 140 specialists in over 20 countries) under one Notified Body system.
In 2021, DNV submitted its application for In-vitro Diagnostic Devices Regulation (EU) 2017/746 (IVDR) to provide a global service for this rapidly growing sector. The application process is in its final stages.
*Note: This MDR readiness self-assessment checklist contains selected MDR requirements to be addressed by the manufacturer which are in addition to those defined in ISO 13485:2016. The purpose is to enable manufacturers to perform a readiness self-assessment of their quality management system. This checklist has been developed as a free tool for the purpose of making a readiness evaluation of a quality system and not for determining or guaranteeing compliance to the MDR.
More information
Factsheet for Manufacturers
Medical devices change of legislation, find out what you need to know if you are a manufacturer.
Step by Step Guide for Manufacturers
Implementation model for medical devices regulation, download the step by step guide.
Factsheet for Authorised Representatives, Importers and Distributors.
For authorised representatives, importers and distributors of medical devices and in vitro diagnostic medical devices.
Factsheet for Procurement Ecosystem
For people within hospitals, clinics (or associations of clinics), ministries and competent bodies responsible for the procurement of medical devices and in vitro diagnostic medical devices.
Factsheet for Authorities in non-EU/EEA States
For regulatory/competent authorities in countries that are not part of the EU/EEA area.
Frequently Asked Questions
| Answer |
|---|
AnswerDNV GL Presafe AS (Notified Body 2460) is a Notified Body for EU Medical Devices Regulation (MDR) 2017/745.
|
| Answer |
|---|
AnswerThe following fee structure applies for conformity assessment activities:
|
| Answer |
|---|
AnswerDownload the process and requirements document here.
|